Why Ultrasound?
Ultrasound is a safe, non-invasive technology that can both see inside the body and precisely deliver energy to targeted tissues. Unlike many medical tools, it can support every stage of an intervention, from diagnosis to treatment to real-time monitoring.
The Keller Lab works at this intersection. We develop new imaging approaches for diagnosis, design adaptive focused ultrasound therapies for diverse disease targets, and build feedback-controlled systems that integrate imaging and therapy into unified, image-guided interventions


Current Research Projects

Novel contrast agents and contrast imaging
Contrast-enhanced ultrasound (CEUS) enables real-time visualization of blood perfusion without the use of ionizing radiation. When exposed to ultrasound, contrast agents (microbubbles) oscillate, generating nonlinear signals for imaging and, at higher acoustic pressures, producing micromechanical forces that can be harnessed for therapeutic applications. Our group develops novel contrast agents for imaging and therapy, and new methodologies for improved contrast imaging, including super-resolution imaging of the microvasculature.

Cavitation dynamics and control
When microbubbles are exposed to ultrasound, they emit signals that can be localized and interpreted to monitor therapeutic efficacy. Our group develops image-guided intervention frameworks that interleave ultrasound contrast imaging, cavitation-mediated therapeutic ultrasound, and cavitation imaging within a single automated, user-independent control system.

Drug delivery and antibiofilm therapies
Ultrasound-mediated cavitation can transiently modify biological barriers to enhance the transport and localization of therapeutics. This capability has broad relevance across applications ranging from solid tumors, where dense extracellular matrices limit drug penetration, to bacterial biofilms, whose physical structure and altered phenotypes protect microbes from antimicrobial agents. While cavitation-mediated ultrasound shows promise for improving drug delivery and biofilm disruption, it remains unclear how maximally effective parameters influence host immune responses or whether displaced cells (tumor or bacterial) are effectively cleared or persist to drive disease progression. Our group develops physiologically relevant models to study host responses to cavitation-mediated therapies for enhanced drug delivery and biofilm control.

Probing ultrasound-induced mechanotransduction
The use of focused ultrasound opens avenues to non-invasively stimulate mechanosensitive cell types, like neurons, osteocytes/chondrocytes, and immune cells. To further explore these mechanisms, our group evaluates ultrasound parameters that induce a variety of biomechanical forces with or without cavitation nuclei (such as shear force, compression, radiation force, or cavitation-induced microjetting) and assess downstream functional cellular responses, including changes in gene expression and phenotype.
Featured Publications
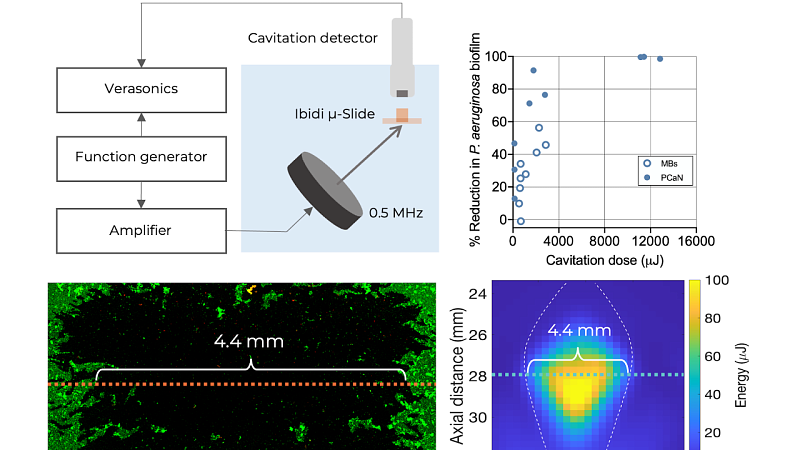
2024
Quantitative evaluation of anti-biofilm cavitation activity seeded from microbubbles or protein cavitation nuclei by passive acoustic mapping
Physics in Medicine and Biology
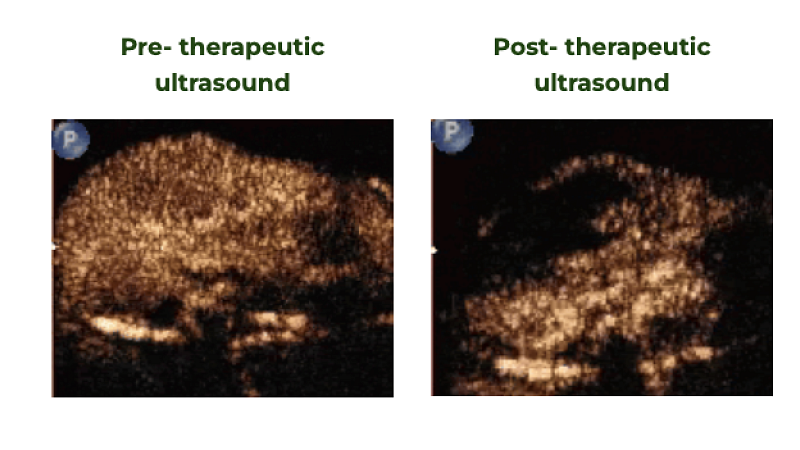
2019
Image-guided treatment of primary liver cancer in mice leads to vascular disruption and increased drug penetration
Ultrasound in Medicine and Biology
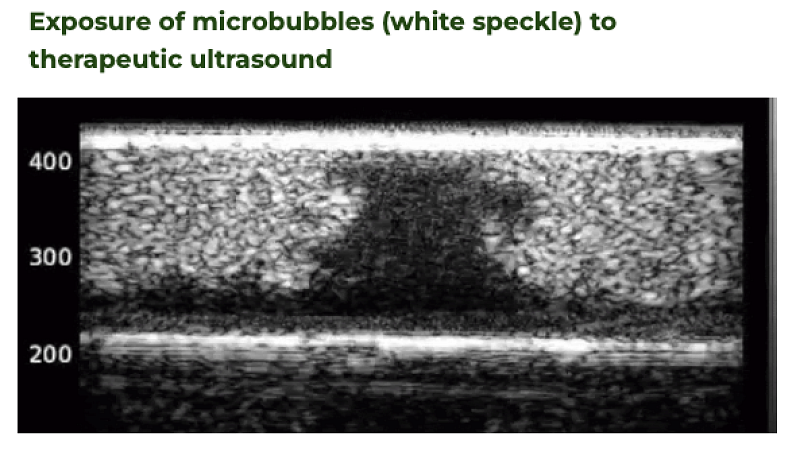
2019
Ultrasound imaging of microbubble activity during sonoporation pulse sequences
Ultrasound in Medicine and Biology
