Department of Bioengineering
The Benoit Lab
Therapeutic Biomaterials
Current Projects
01
Developing Targeted Drug Delivery Systems to Treat Leukemia
Acute myeloid leukemia (AML) recurrences are attributed to leukemia stem cells (LSCs) and a harsh marrow microenvironment that supports AML cell survival. LSCs in particular survive conventional chemotherapy and radiation treatments leading to relapse in patients. Drugs such as parthenolide (PTL) and micheliolide have has shown remarkable efficacy in inducing selective apoptosis in LSCs. However, these compounds' low water solubility prevents them from reaching therapeutically effective levels in the blood stream. To circumvent this problem, we are developing a novel micelle delivery system to solubilize and target compounds. The micelles are formed from diblock copolymers that are hydrophilic and hydrophobic. In physiologic solutions, the carriers self-assemble into spherical carriers with hydrophobic interiors that can be loaded with hydrophobic compounds. Thus, we hypothesize that these carriers will greatly increase compound blood concentrations, enhancing their chemotherapeutic efficacy. We also aim to conjugate peptides to the corona of the micelles to home to the bone microenvironment and the LSCs, reducing the overall physiologic burden of drug and to selectively ablate LSCs, reducing AML recurrence rates. Figure 1 shows a schematic of the development of bone marrow (BM) and LSC targeted drug delivery systems (DDS) for acute myeloid leukemia (AML). Briefly, to maximize BM drug accumulation, a targeting peptide with specificity towards osteoclast deposited tartrate-resistant acid phosphatase (TRAP) was introduced to polymeric nanoparticles (NP) to rescue the bone marrow microenvironment (BMME) through c-chemokine ligand 3 (CCL3) disruption and LSC inhibition. The bone-targeted NP system is broadly applicable to bone and bone marrow associated diseases including multiple myeloma, bone regeneration, osteomyelitis, etc. Regardless of the system chosen, these studies show the potential of targeted DDS to increase tissue/cell-specific drug delivery and reduce off-target effects and toxicities associated with traditional treatment options.
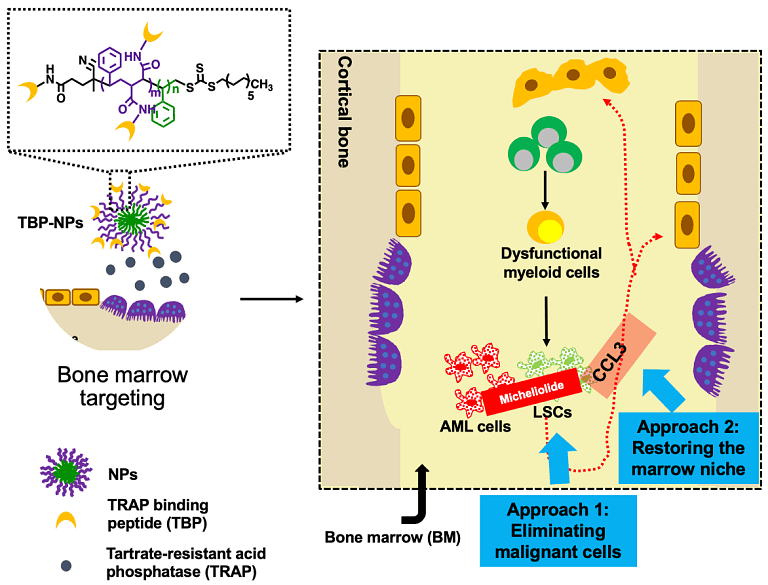
Project Collaborators: Rudi Fasan, Benjamin Frisch, Craig Jordan
02
Developing Tissue Mimetics of the Outer Retinal Blood Barrier
Dysfunction of the outer retinal blood barrier (oRBB), is implicated in age-related macular degeneration (AMD), the leading cause for blindness in industrialized countries. Despite its prevalence, the mechanisms of AMD remain poorly understood. Animal models for AMD are severely limited due to physiological differences and impracticality. Advances in tissue engineering and organ-on-a-chip technology have catalyzed the rise of microphysiological systems (MPS) as an alternative for preclinical testing. Thus, to overcome this major hurdle, the goal of this project is to engineer improved MPS to model the outer retinal blood barrier using engineered hydrogel materials, nanoporous silicon nitride membranes, induced pluripotent stem cells, and microfluidic devices. This research will establish an in vitro platform for interrogating retinal (patho)physiology and investigating the mechanistic complexities of the oRBB microenvironment.
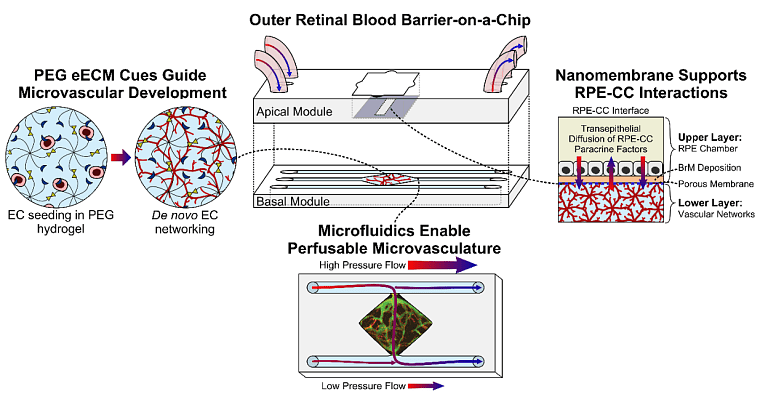
Project Collaborators: Ruchira Singh, Jinjiang Pang, Jim McGrath, Lisa DeLouise
03
Hydrogels for Local Delivery of Therapeutics
Hydrogels can be exploited to encapsulate and deliver cells and biomolecules therapeutically, as delivery characteristics can be intimately controlled through alterations in hydrogel biophysical and biochemical structure. We use poly(ethylene glycol) (PEG) hydrogels as local delivery systems for cells and/or therapeutics that act locally to aid tissue regeneration in several applications, including ischemic tissue disorders, fractures, and critical-size bone defects. We hypothesize that hydrogels designed to encapsulate cells and control the availability of cellularly-released paracrine factors and/or other therapeutic molecules such as growth factors, gene vectors, or small interfering RNA, will be useful for these therapeutic strategies. Our highly tunable PEG hydrogel platform can encapsulate soluble factors for diffusion-controlled release and/or support cell-mediated release by covalent conjugation of therapeutics via enzymatically degradable linkers. For example, peptide mimetics of growth factors, such as NGF or VEGF, can be tethered to the polymer using matrix metalloproteinase (MMP)-cleavable linkers to create cell-responsive delivery systems.
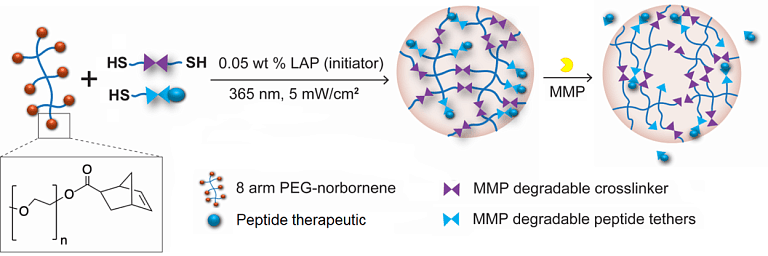
Project Collaborators: Hani Awad, Jared Mereness, Lisa DeLouise
04
pH-Responsive Nanoparticle Drug Delivery to Control Oral Biofilm Virulence
Tooth decay, often referred to as cavities and known clinically as dental caries, affects over 60% of children and nearly all adults worldwide and costs billions of dollars annually. Each year, dentists across the nation fill more than 200 million cavities! Tooth decay is a disease caused by bacteria that reside in the human mouth. These bugs live on tooth and gum surfaces within the mouth and can build up to create what is known as an oral biofilm, commonly called dental plaque. This plaque can be quite destructive due to the acids produced as bacteria digest sugars found in the human diet. These acids dissolve the enamel surface covering the teeth, and create small holes that grow to become cavities. Despite this knowledge, current treatment options using topically applied drugs offer little to no protection. This fact is due to the poor solubility or salivary clearance of drugs as well as a protective barrier created by the biofilm and results in insufficient contact time to kill bacteria on the tooth surface. Therefore, we are interested in killing these bacteria and preventing the biofilms before they become destructive by leveraging key features of tooth decay pathology, such as the stark drop in pH, in the fight against the bacteria. Our lab works with a team of collaborators to design and test drug delivery systems capable of penetrating dental plaque and releasing antibacterial drugs precisely where cavities develop: the acid-covered tooth surface. Specifically, we've developed polymer nanoparticles that can load poorly soluble drugs and then release those drugs due to changes in local pH conditions, such as the acidic environments created by oral biofilms (see Figure 2). In this way, we can deliver the right drug, to the right place, at the right time to inhibit biofilm formation and thus save teeth from the harsh acids that cause decay.
Project Collaborators: Hyun 'Michel' Koo, Marlise Klein, Jin Xiao, Anne Meyer, Karen Guillemin
05
Radioprotection and Regeneration of Salivary Glands
For over 50,000 U.S. patients annually diagnosed with head and neck cancers, radiation therapy frequently causes severe salivary gland dysfunction (xerostomia). To address this, we are pursuing multiple complementary strategies. First, we developed a radioprotective approach using siRNA-nanoparticle complexes to silence genes associated with radiation damage, achieving remarkable preservation of salivary gland histology and function. Second, we are developing hydrogel microenvironments to study salivary gland cells and enable transplantation-based structural and functional rescue of atrophic and irradiated glands. Finally, to overcome the limitations of current in vitro models — which fail to maintain secretory acinar cell phenotype — we are building functional human salivary gland tissue chips using a high-throughput microbubble (MB) array platform, where micron-scale spherical cavities promote cell-cell contact and tissue assembly in geometries mimicking native gland architecture. Together, these tools will dramatically advance our ability to develop and evaluate radioprotective and regenerative therapeutic strategies.
Project Collaborators: Catherine Ovitt, Lisa DeLouise, Melinda Larsen, Paul Dunman, Jared Mereness
06
Targeted Drug Delivery Systems for Musculoskeletal Applications
Our laboratory leverages RAFT polymerization to synthesize well-controlled polymeric nanoparticles (NPs) for targeted, spatially- and temporally-controlled delivery of small molecule drugs and siRNA to musculoskeletal tissues. A key application of this platform is treating non-union fractures, which affect ~10% of the 15 million Americans who suffer fractures annually. Current pharmacologic options like BMP-2/7 and Wnt/β-catenin agonists are limited by off-target biodistribution and safety concerns at the doses needed to reach fracture sites. To address this, we developed a fracture-targeted NP system functionalized with a peptide that binds TRAP, a protein deposited by osteoclasts at injury sites, achieving preferential accumulation at fractures, enhanced local β-catenin activation, expedited callus formation, and 2.5–4-fold improvements in mechanical strength versus controls. We are now working to optimize peptide targeting, establish therapeutic dosing windows, and validate this approach in clinically-relevant non-union models.
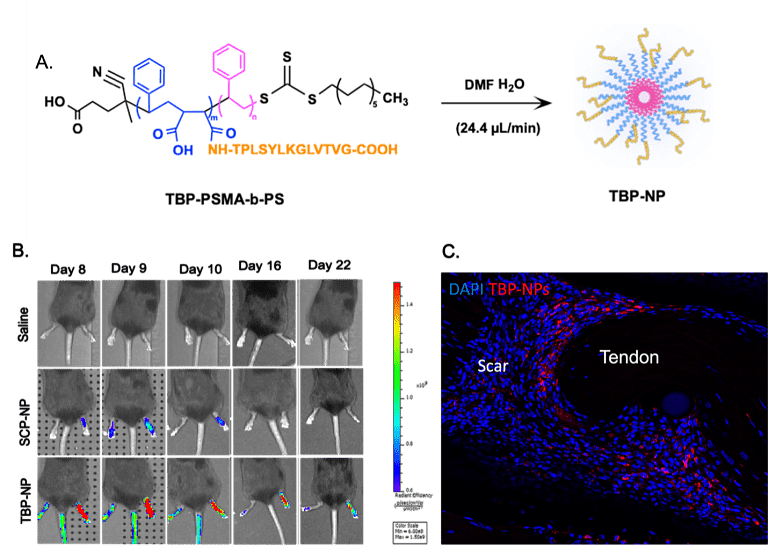
Project Collaborators: Edward Puzas, Alayna Loiselle, Nick Willett
07
Tissue Engineered Periosteum Approaches to Heal Bone Allograft Transplants
Although most orthopaedic fractures heal, the clinical management of critical (>3mm) segmental defects continues to present major challenges for both amputation and limb salvage approaches. The gold standard for treatment of these defects is allografts, bone grafts from cadaveric donors. While most fractures and autografts fully heal, the healing capacity of allografts is severely limited, as the periosteum plays an essential role in the healing process of both fractures and autografts. Periosteal stem cells have been shown to be necessary for the induction of robust endochondral and intramembraneous bone formation, essential for effective healing, and neovascularization of autografts. However, allografts are processed such that there are no viable stem cells or periosteum remaining. Therefore, by providing a pseudo-periosteum to revitalize allografts, a similar robust healing response may be induced. We aim to exploit hydrogel cell/drug delivery vehicles as a tissue-engineered periosteum, evaluating different delivery regimes and hydrogel biochemical properties to promote stem cell proliferation and differentiation in vitro and in vivo within a murine segmental defect model. Our global hypothesis is that a tissue-engineered periosteum will promote allograft integration and healing thus reducing allograft failure rates.
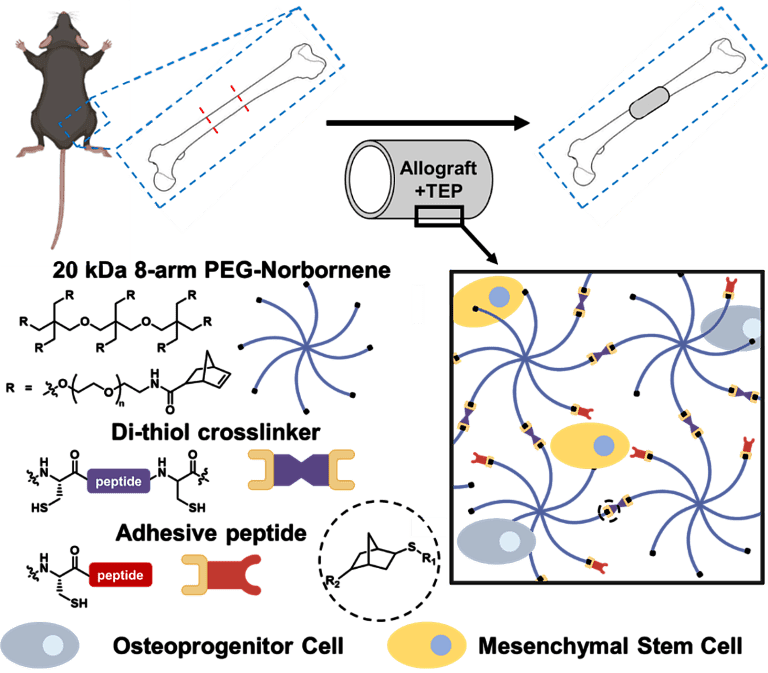
Project Collaborators: Regine Choe, Roman Eliseev, Xinping Zhang
08
Promoting Regenerative Tendon Healing Using Materials Design Strategies
Tendon injuries affect over 15 million people annually, often resulting in chronic pain and limited function due to fibrotic, scar-forming healing rather than true tissue regeneration. To address this, we are developing regenerative strategies through engineered extracellular matrices (eECMs) that incorporate both biochemical and mechanical cues. On the biochemical side, we are working with collaborators in the Loiselle lab to characterize an enhanced tendon healing environment in a novel murine model, with the goal of identifying key differences from fibrotic healing and using these insights to design eECMs that recapitulate regenerative conditions. On the mechanical side, given that tendons naturally experience large mechanical stresses, we are designing hydrogel eECMs capable of promoting anisotropic tenocyte alignment and on-demand mechanical actuation to simulate rehabilitative loading — hypothesized to restore function more effectively than static, isotropic materials.
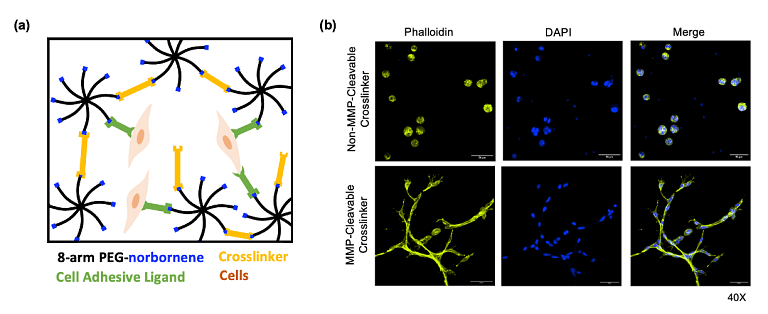
Project Collaborators: Alayna Loiselle, Tayler Hebner
Featured Publications
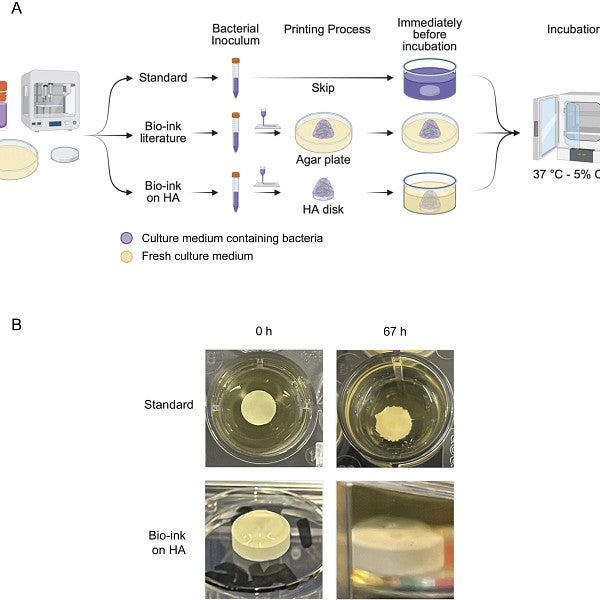
2025
Reproducible 3D bioprinting of Streptococcus mutans to create model oral biofilms
Microbiology Spectrum

2025
Salivary gland tissue chip screening identifies candidate radioprotective drugs
Communications Medicine
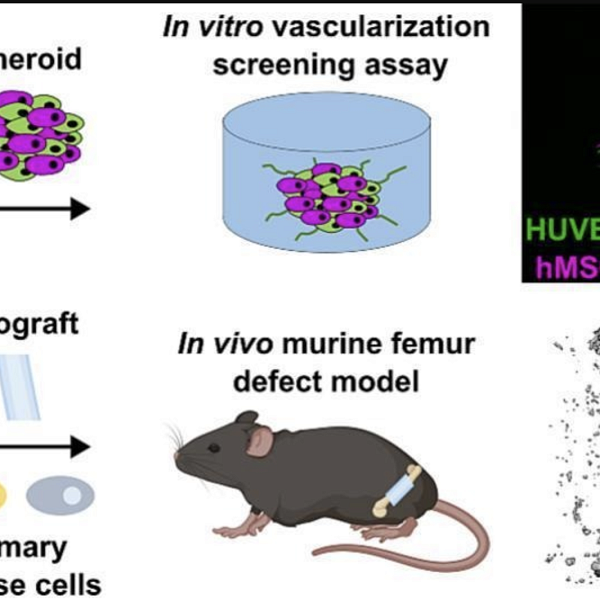
2025
Leveraging the predictive power of a 3D in vitro vascularization screening assay for hydrogel-based tissue-engineered periosteum allograft healing
Biomaterials Advances
The Benoit Lab
In 2022, the Benoit lab relocated to the University of Oregon's Phil and Penny Knight Campus for Accelerating Scientific Impact. Based in the Department of Bioengineering in Eugene, Oregon, the Benoit lab develops therapeutic biomaterials to treat diseases, control cell behavior, and answer fundamental biological questions.
